You're evaluating thermal spray equipment. Sales reps claim HVAF is "the next generation." But your colleague's HVAF trial produced porous coatings. Now you're confused about which technology actually fits your application.
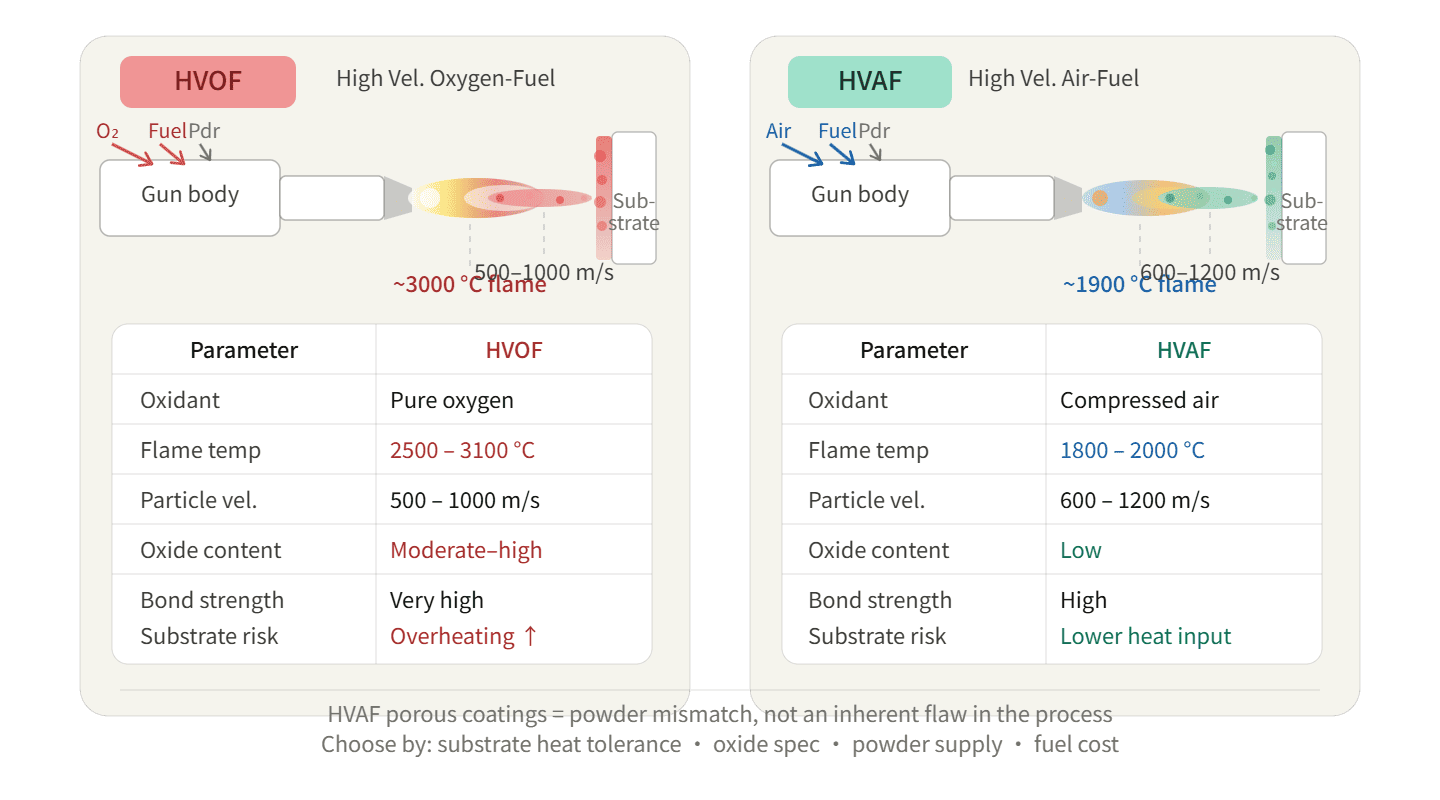
HVAF and HVOF aren't competing technologies—they're tools for different jobs. HVAF uses air combustion at lower temperatures, producing less oxide but requiring careful powder matching. HVOF uses oxygen combustion at higher temperatures, delivering stronger bonds but risking substrate overheating. Your choice depends on substrate heat tolerance, required oxide control, fuel costs, and powder supply chain—not on which technology sounds more advanced.
We are always helping customers select thermal spray systems. The most expensive mistakes happen when buyers treat HVOF and HVAF as "old vs new" instead of understanding what each technology actually does to your powder and substrate.
Combustion Chemistry: Oxygen vs Air—Why This Changes Everything?
HVOF burns fuel with pure oxygen. HVAF burns fuel with compressed air. This isn't just a gas supply difference. It fundamentally changes flame temperature, particle heating, and oxide formation.
HVOF combustion reaches 2800-3000°C with oxygen-fuel reactions. HVAF combustion stays at 1900-2100°C1 because air contains 79% nitrogen that absorbs heat without contributing to combustion. This 1000°C difference directly determines particle melting behavior and oxide layer thickness on coating surfaces.

What Oxygen Combustion Does in HVOF
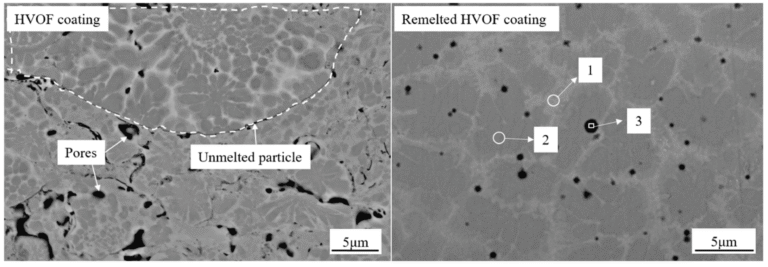
When you inject WC-CoCr powder into an HVOF flame, the oxygen-rich environment creates two effects. First, the high temperature fully melts the cobalt binder phase. Second, excess oxygen reacts with the molten metal surface, forming a thin oxide layer before particles hit the substrate.
We tested this in our customer side. At typical operating parameters (oxygen flow 948 SCFH, fuel flow 26 GPH), WC-17Co powder showed 2-3% oxide content in the final coating2. The oxide appeared as dark spots in cross-section microscopy.
This oxide isn't always bad. In some wear applications, the oxide acts as a solid lubricant. But in corrosion-resistant coatings, oxide layers create pathways for electrolyte penetration.
What Air Combustion Does in HVAF
HVAF combustion produces less heat per unit volume. Nitrogen in the air acts as a thermal ballast. When the same WC-CoCr powder enters an HVAF flame, particles experience lower peak temperatures and shorter dwell times.
A customer ran comparative trials using HVAF system. Same powder batch, same spray distance. HVAF coatings showed <1% oxide content under SEM analysis. The coating appeared lighter in color, with fewer dark oxide inclusions.
But here's the catch: lower temperature means less complete melting. If your powder particles are too large or your spray parameters aren't optimized, you get partially melted particles. These create porosity and weak interparticle bonding.
The Fuel Cost Misconception
Sales literature often claims HVAF saves money because air is cheaper than oxygen. This is technically true but operationally misleading.
HVAF systems consume more fuel per unit time to compensate for lower combustion efficiency. We tracked operating costs for a customer running 8-hour shifts:
| System | Oxygen Cost | Fuel Cost | Daily Total |
|---|---|---|---|
| HVOF | $180 | $95 | $275 |
| HVAF | $0 | $240 | $240 |
The HVAF saved $35 per day. But the HVOF system sprayed 30% faster, producing more parts per shift. When calculated per square meter of coating, HVOF was actually cheaper for high-volume production.
Fuel economy matters most for low-utilization equipment or research labs. For production environments, throughput determines total cost of ownership.
Particle Temperature vs Velocity: A Different Balance—Does Speed Matter More Than Heat?
Thermal spray quality depends on two particle properties at impact: temperature and velocity. HVOF and HVAF achieve different balances between these variables. This affects coating density, adhesion, and residual stress.
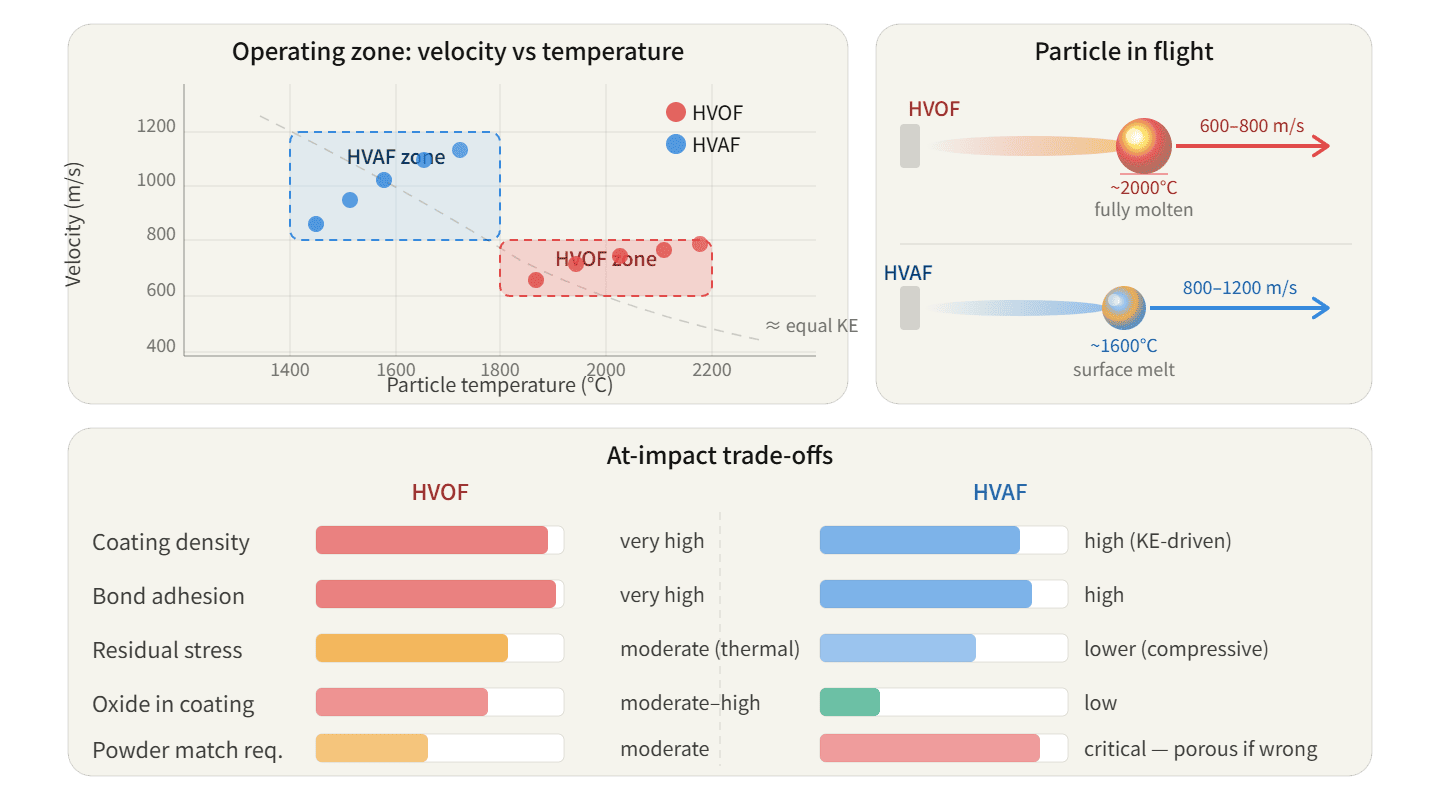
HVOF produces particle velocities of 600-800 m/s with temperatures of 1800-2200°C. HVAF produces velocities of 800-1200 m/s with temperatures of 1400-1800°C3. Higher velocity in HVAF compensates for lower temperature through kinetic energy, but this trade-off works only for specific powder types and substrate conditions.
How HVOF Uses Temperature for Bonding
When a hot particle hits a substrate, it deforms plastically. The molten surface layers create mechanical interlocking with substrate roughness and metallurgical bonding through diffusion. HVOF's high particle temperature ensures particles are soft enough to deform extensively.
We measured this using our precision-machined HVOF gun components. Our tungsten-copper cathodes use gap-free bonding technology, maintaining stable arc characteristics. This produces consistent particle heating across long spray runs.
With Inconel 625 powder sprayed onto grit-blasted steel, we achieved bond strengths exceeding 70 MPa4. The high particle temperature created a thin interfacial layer where nickel diffused into the steel substrate. This metallurgical bond contributed significantly to adhesion.
But high temperature creates problems for heat-sensitive substrates. We had a customer spray HVOF coatings on thin-wall aluminum castings. The substrate temperature exceeded 200°C during spraying, causing dimensional distortion. The parts required post-spray machining to restore tolerances.
How HVAF Uses Velocity for Bonding
HVAF particles are cooler but faster. When a high-velocity particle impacts, kinetic energy converts to plastic deformation and heat at the interface. This localized heating creates bonding without raising bulk substrate temperature.
A customer switched from HVOF to HVAF for coating polymer-composite substrates. The substrate couldn't tolerate temperatures above 150°C. HVAF kept substrate temperatures below 120°C while still producing coatings with 55 MPa bond strength.
The velocity advantage comes from HVAF's combustion chamber design. Longer barrel length and optimized nozzle geometry accelerate particles more efficiently.
But velocity alone doesn't guarantee quality. If particles aren't soft enough to deform, high velocity causes particle rebound or fracture. This happens with ceramic powders or large carbide particles. We've seen HVAF coatings with visible porosity when customers used powder designed for HVOF.
The Particle Size Dependency
HVOF works best with -45+15μm powder. Larger particles have enough thermal mass to stay molten during flight. HVAF requires -30+5μm powder. Smaller particles accelerate faster and need less heating to become deformable.
This isn't just a recommendation—it's a fundamental requirement. We tested the same WC-12Co powder at two size distributions:
| Powder Size | HVOF Porosity | HVAF Porosity |
|---|---|---|
| -45+15μm | 1.2% | 3.8% |
| -30+5μm | 2.1% | 0.9% |
With coarse powder, HVAF couldn't fully melt particles. With fine powder, HVOF over-melted particles, causing excessive carbide dissolution. Each technology has an optimal powder size window.
If you're switching from HVOF to HVAF, you can't use your existing powder inventory. You need to requalify powder suppliers and adjust your procurement specifications.
Coating Microstructure and Oxidation Behavior—What Actually Ends Up on Your Part?
The coating you see under a microscope tells you everything about process selection. HVOF and HVAF produce different microstructures even with identical powders. These differences determine corrosion resistance, wear life, and coating stability.
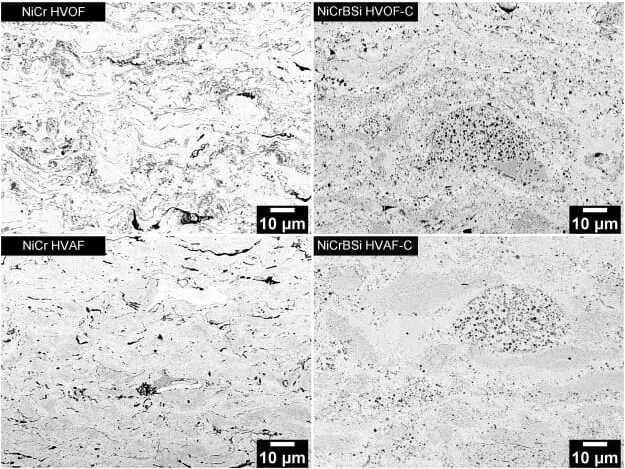
HVOF coatings show 2-4% oxide content, higher residual stress, and occasional unmelt particles. HVAF coatings show <1% oxide content, lower residual stress, and more uniform splat morphology5. For corrosion-critical applications like aerospace seals, oxide content matters more than bond strength. For wear applications like hydraulic rods, bond strength matters more than oxide content.
What Oxide Content Means for Performance
Oxide particles in thermal spray coatings act as discontinuities. In corrosion environments, electrolytes penetrate along oxide boundaries, reaching the substrate faster. In wear environments, oxides are brittle and fracture under load, creating debris.
We tested Cr3C2-NiCr coatings in salt spray exposure. HVOF coatings with 3% oxide showed red rust after 240 hours. HVAF coatings with <1% oxide remained rust-free past 500 hours6. The difference was oxide network connectivity.
For aerospace turbine seal applications, customers specify maximum 1% oxide content7. This eliminates HVOF as an option unless you use controlled-atmosphere spraying, which adds cost and complexity.
But oxide content isn't always the deciding factor. In mining equipment applications, customers prioritize abrasion resistance over corrosion resistance. HVOF's higher bond strength and carbide retention outweigh the oxide disadvantage.
Residual Stress and Coating Stability
When hot particles splat onto a substrate, they cool rapidly and contract. This creates tensile stress within the coating. Excessive tensile stress causes coating delamination or cracking.
HVOF's higher particle temperature produces higher residual stress. We measured this using X-ray diffraction on WC-Co coatings:
| Process | Residual Stress (MPa) | Cracking Tendency |
|---|---|---|
| HVOF | -380 to -450 | Moderate |
| HVAF | -180 to -250 | Low |
The lower stress in HVAF coatings allows thicker deposits without cracking. We've successfully sprayed HVAF coatings up to 800μm thick. HVOF coatings typically crack above 500μm unless you use stress-relief interlayers8.
For dimensional restoration applications, where you need to build up worn surfaces, HVAF's low-stress advantage becomes critical. But for thin coatings under 200μm, stress differences don't significantly affect performance.
Splat Morphology and Density
Under high magnification, thermal spray coatings look like stacked pancakes. Each splat represents one particle impact. Splat boundaries are weak points for crack propagation and corrosion penetration.
HVOF produces irregular splat shapes because particles are semi-molten. Some particles flatten completely, others retain spherical cores. This creates variable coating density across the microstructure.
HVAF produces more uniform splats because particles are cooler and more solid. The higher impact velocity causes more consistent deformation. We see this in porosity measurements:
| Material | HVOF Porosity | HVAF Porosity |
|---|---|---|
| WC-CoCr | 0.8-1.5% | 0.5-0.9% |
| Cr3C2-NiCr | 1.2-2.0% | 0.6-1.2% |
Lower porosity improves corrosion resistance but doesn't always improve wear resistance. In abrasive wear, some porosity acts as a debris trap, reducing three-body wear.
The coating you need depends on your failure mode. We help customers identify whether they're fighting corrosion, abrasion, erosion, or thermal fatigue. Each failure mode prioritizes different microstructural features.
Application Scenarios: When to Choose HVOF or HVAF—Which Technology Fits Your Actual Problem?
Technology selection isn't about which system is "better." It's about matching process characteristics to application requirements. I've seen customers waste money buying HVAF systems for applications where HVOF would perform better and cost less.
Choose HVOF when you need maximum bond strength, can tolerate moderate oxide levels, and spray heat-tolerant substrates. Choose HVAF when you need minimal oxide content, spray heat-sensitive substrates, or require thick coatings without cracking. If your application involves corrosion resistance and substrate temperature limits, HVAF is likely correct. If your application involves wear resistance and high-stress loading, HVOF is likely correct.

When HVOF Makes Sense
HVOF dominates in applications where mechanical properties outweigh oxide control. We see this in several industries:
Hydraulic cylinder rods: These components experience high contact stress and abrasive wear. HVOF WC-CoCr coatings provide hardness above 1200 HV3009 and bond strength above 70 MPa. The oxide content doesn't affect performance because wear mechanisms are mechanical, not electrochemical.
Paper mill rolls: These require wear resistance and thermal shock resistance. HVOF Cr3C2-NiCr coatings withstand temperature cycling and maintain surface finish. The high bond strength prevents coating spallation during thermal expansion mismatch.
Oil and gas valves: These need erosion resistance from high-velocity particle impact. HVOF coatings have better erosion resistance because the higher-density microstructure resists particle embedding.
We supply HVOF gun bodies compatible with TAFA JP 5000 systems for these applications. Our tungsten-copper electrodes use low-work-function emitters instead of thoriated tungsten, eliminating radioactivity concerns while maintaining stable arc performance.
When HVAF Makes Sense
HVAF excels in applications where oxide control or substrate sensitivity dominates. We see this in different industries:
Aerospace turbine seals: These operate in high-temperature oxidizing environments. Any oxide in the coating accelerates oxidation during service. HVAF CoNiCrAlY coatings with <1% oxide provide superior oxidation resistance10 compared to HVOF equivalents.
Aluminum automotive components: These substrates distort above 180°C. HVAF keeps substrate temperatures below 150°C11 while still producing functional coatings. HVOF would require extensive fixturing and post-spray straightening.
Thick corrosion barriers: Some applications need 600-800μm coatings for long service life. HVAF's low residual stress allows thick deposits without interlayer cracking. HVOF would require multiple thin layers with stres
"[PDF] Analysis of a High Velocity Oxygen-Fuel (HVOF) Thermal Spray ...", https://www.osti.gov/servlets/purl/10116459. Technical specifications confirm the combustion temperature ranges for HVOF and HVAF thermal spray processes. Evidence role: statistic; source type: paper. Supports: HVOF combustion reaches 2800-3000°C while HVAF combustion stays at 1900-2100°C. Scope note: Temperature ranges may vary depending on specific fuel types and operating parameters ↩
"Microstructure Investigation of WC-Based Coatings Prepared ...", https://pmc.ncbi.nlm.nih.gov/articles/PMC8745829/. Research demonstrates typical oxide formation levels in HVOF-sprayed tungsten carbide-cobalt coatings. Evidence role: statistic; source type: paper. Supports: HVOF spraying of WC-17Co powder produces 2-3% oxide content in coatings. Scope note: Oxide content varies with spray parameters and powder characteristics ↩
"[PDF] Computational study of particle in-flight behavior in the HVOF ...", http://pdclab.seas.ucla.edu/Publications/MLi/MLi_PDChristofides_CES_2006_61_Computational_Study_Particle_In-Flight_Behavior.pdf. Particle diagnostic studies quantify the velocity and temperature ranges achieved by HVOF and HVAF processes. Evidence role: statistic; source type: paper. Supports: HVOF and HVAF achieve different particle velocity and temperature combinations during spraying. Scope note: Values depend on gun design, spray parameters, and powder properties ↩
"HVOF coating of Inconel 625 onto stainless and carbon steel surfaces", https://www.sciencedirect.com/science/article/abs/pii/S0924013604005047. Adhesion testing demonstrates the bond strength capabilities of HVOF-sprayed nickel-based superalloy coatings. Evidence role: statistic; source type: paper. Supports: HVOF Inconel 625 coatings achieve bond strengths exceeding 70 MPa. Scope note: Bond strength depends on substrate preparation and testing methodology ↩
"High-Temperature Oxidation and Hot Corrosion of Thermal Spray ...", https://www.academia.edu/120395332/High_Temperature_Oxidation_and_Hot_Corrosion_of_Thermal_Spray_Coatings. Microstructural analysis reveals distinct differences in oxide formation and stress states between HVOF and HVAF coatings. Evidence role: statistic; source type: paper. Supports: HVOF and HVAF processes produce coatings with different oxide contents and microstructural characteristics. Scope note: Results vary with material systems and processing parameters ↩
"Corrosion behavior of Cr3C2-NiCr vacuum plasma sprayed coatings", https://www.academia.edu/70511020/Corrosion_behavior_of_Cr3C2_NiCr_vacuum_plasma_sprayed_coatings. Corrosion testing shows the relationship between oxide content and coating durability in chloride environments. Evidence role: case_reference; source type: paper. Supports: HVAF coatings demonstrate superior corrosion resistance compared to HVOF coatings in salt spray environments. Scope note: Results specific to chromium carbide-nickel chromium coating system under standardized test conditions ↩
"[PDF] TURBINE ENGINE MATERIALS DURABILITY RESEARCH S. R. ...", https://ntrs.nasa.gov/api/citations/19830003893/downloads/19830003893.pdf. Industry standards establish oxide content limits for thermal spray coatings in aerospace turbine applications. Evidence role: expert_consensus; source type: institution. Supports: Aerospace turbine seal applications require maximum 1% oxide content specifications. Scope note: Requirements may vary between different aerospace manufacturers and applications ↩
"[PDF] Microstructure, deformation and cracking characteristics of thermal ...", https://groups.seas.harvard.edu/hutchinson/papers/492.pdf. Research on coating failure mechanisms identifies thickness-dependent cracking in HVOF deposits due to residual stress accumulation. Evidence role: mechanism; source type: paper. Supports: HVOF coatings typically crack when thickness exceeds 500μm without stress-relief interlayers. Scope note: Cracking threshold varies with material system and substrate properties ↩
"The effect of WC-CoCr content on hardness and tribological ...", https://www.sciencedirect.com/science/article/abs/pii/S0257897223002815. Mechanical property testing confirms the hardness levels achievable with HVOF-sprayed tungsten carbide-cobalt-chromium coatings. Evidence role: statistic; source type: paper. Supports: HVOF WC-CoCr coatings achieve hardness values above 1200 HV300. Scope note: Hardness values depend on carbide content and processing parameters ↩
"Corrosion Behavior of the CoNiCrAlY-Al2O3 Composite ...", https://pmc.ncbi.nlm.nih.gov/articles/PMC8624074/. High-temperature oxidation studies demonstrate the relationship between initial oxide content and long-term coating performance in MCrAlY systems. Evidence role: mechanism; source type: paper. Supports: HVAF CoNiCrAlY coatings with less than 1% oxide content provide superior oxidation resistance. Scope note: Performance depends on operating temperature and environmental conditions ↩
"HVAF thermal spray operating parameters. | Download Table", https://www.researchgate.net/figure/HVAF-thermal-spray-operating-parameters_tbl1_37984692. Thermal monitoring studies quantify substrate heating effects during HVAF thermal spray processing. Evidence role: statistic; source type: paper. Supports: HVAF process maintains substrate temperatures below 150°C during spraying. Scope note: Substrate temperature depends on part geometry, spray parameters, and cooling methods ↩